|
This work expands the realm of possibility for uses of PC from anti-fogging lenses to durable biological devices, allowing scientists and engineers to take advantage of the many attractive physical properties of PC without limitations of hydrophobicity. Using this protocol, the vapor density is calculated by measuring the mass and volume of the vapor. The resulting films decrease PC surface water contact angles (θ) to as low as θ < 10° (superhydrophilic) by continuous growth or sequential extension. Sodium iodide NaI or INa CID 5238 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities. In this lab, students determine the molar mass of the volatile liquids ethanol and 2-propanol using the Dumas method. From these PC-tethered RAFT initiators, hydrophilic polymer brushes are grown under aqueous conditions, visible light, and ambient atmosphere. Chemical formula (Hill notation) Molar mass (g/mol) Modify Clear mnM. A method is developed to immobilize reversible addition–fragmentation chain transfer (RAFT) agents on PC substrates. Select elements and see the molar mass of the compound. This work presents a facile method to chemically modify PC surfaces with superhydrophilic polymer brushes. Though nontoxic and biocompatible, its inherent hydrophobicity limits its potential in applications that require hydrophilicity or use in the body. Iodoform test: It doesn’t react positively.Polycarbonate (PC) is a popular consumer plastic due to its light weight, optical clarity, mechanical strength, and temperature stability. Iodoform test: Ethanol is warmed with iodine in the presence of NaOH, it forms a yellow-colored precipitate. Thus cannot use in beverages.Ĭhemical nature: It is a renewable liquid.Ĭhemical nature: it is a non-renewable liquid. Methanol has a slightly higher level of acid than water. Natural occurrences: Is produced by anaerobic bacteria, phytoplankton, also produced synthetically through catalysis from carbon monoxide, carbon dioxide, and hydrogen. Natural occurrences: This is a by-product of the metabolic process of yeast, commonly be found in overripe fruit, also produced during the germination of many plants 1 g / m o l Hence the correct option is C. Molecular Structure: Ethanol has a linear structure. Molar mass of ethanol (C 2 H 5 O H) 2 (1 2) + 6 (1) + 1 6. Ethanol and dimethyl ether are organic compounds having the same molar mass but different chemical structures. 
The molar mass of isopropanol is 60 g/mol. Molar Mass: The molar mass of ethanol is 46 g/mol. Isopropanol is an alcohol having the chemical formula C 3 H 8 O. Or else, we can use petrochemical processes. Ethanol: C2H5OH: 46.069: Ethene: 28.05: Ethyl Alcohol. Ethanol is an alcohol having the chemical formula C 2 H 5 OH. The key difference between ethanol and propanol is that the ethanol contains two carbon atoms per molecule whereas the propanol contains 3 carbon atoms per. 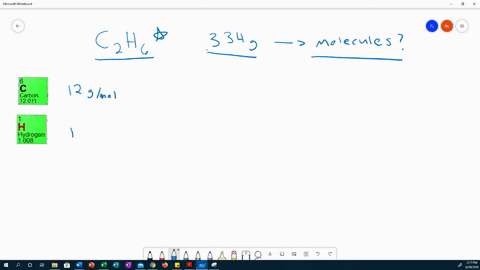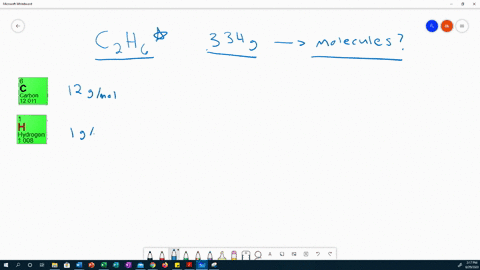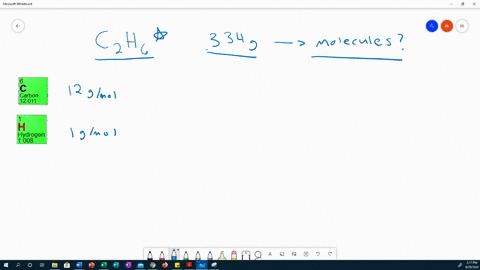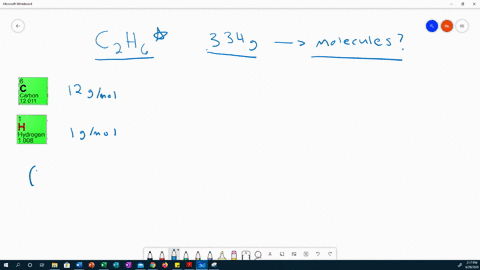
Higher compare to methanol.īoiling point: 64.7☌. The molar mass of a substance, also often called molecular mass or molecular weight (although the definitions are not strictly identical, but it is only sensitive in very defined areas), is the weight of a defined amount of molecules of the substance (a mole) and is expressed in g/mol. It’s a nondrinking type of alcohol(also known as wood alcohol)īoiling point: 78.37☌. Consumption of alcohol is injurious to health. 
It’s used as an ingredient in drinking alcohol (grain alcohol or simple alcohol). Differences between methanol and ethanol S.NoĬhemical structure: Contains two carbon atoms and a hydroxyl group.Ĭhemical structure: Contains one carbon atom and a hydroxyl group. In this work, we study the molar mass dependence of the thermodiffusion of polyethylene oxide at different temperatures in ethanol, water/ethanol mixture (c.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
